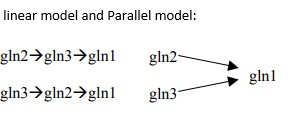
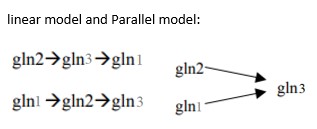
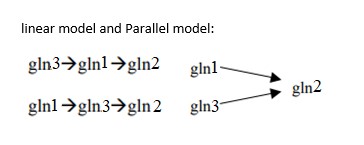
TLS Online TPP Program
More Questions
TLS Online TPP Program
#Question id: 27392
#Unit 1. Molecules and their Interaction Relevant to Biology
Match the following
| Bond | Bond energy in KJ |
| I. C=C | a. 891 |
| II. C ≡ O | b. 946 |
| III. N≡N | c. 1080 |
| IV. C≡N | d. 611 |
TLS Online TPP Program
#Question id: 27393
#Unit 1. Molecules and their Interaction Relevant to Biology
In an exothermic reaction………a…….. and in endothermic reaction …………b………
TLS Online TPP Program
#Question id: 27394
#Unit 1. Molecules and their Interaction Relevant to Biology
Which of the following has smallest size
TLS Online TPP Program
#Question id: 27395
#Unit 1. Molecules and their Interaction Relevant to Biology
Which of the following is the example of polyketides
TLS Online TPP Program
#Question id: 27396
#Unit 1. Molecules and their Interaction Relevant to Biology
Which of the following lipid will have greater melting / boiling point
TLS Online TPP Program
#Question id: 27397
#Unit 1. Molecules and their Interaction Relevant to Biology
Which of the following lipid is precursor for prostaglandin